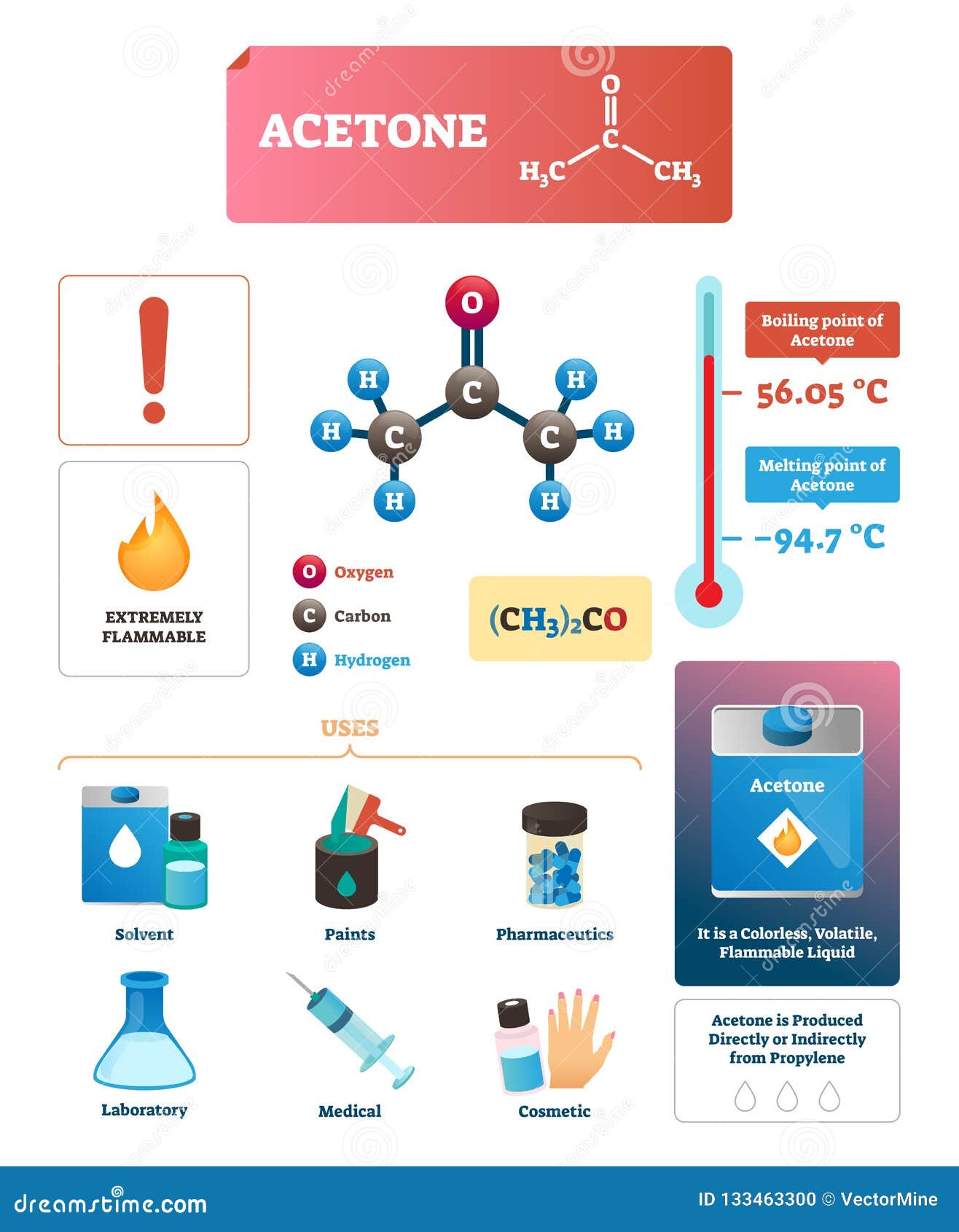
Acetone Vector Illustration. Chemical and Physical Explanation Infographic. Stock Vector - Illustration of educational, medical: 133463300
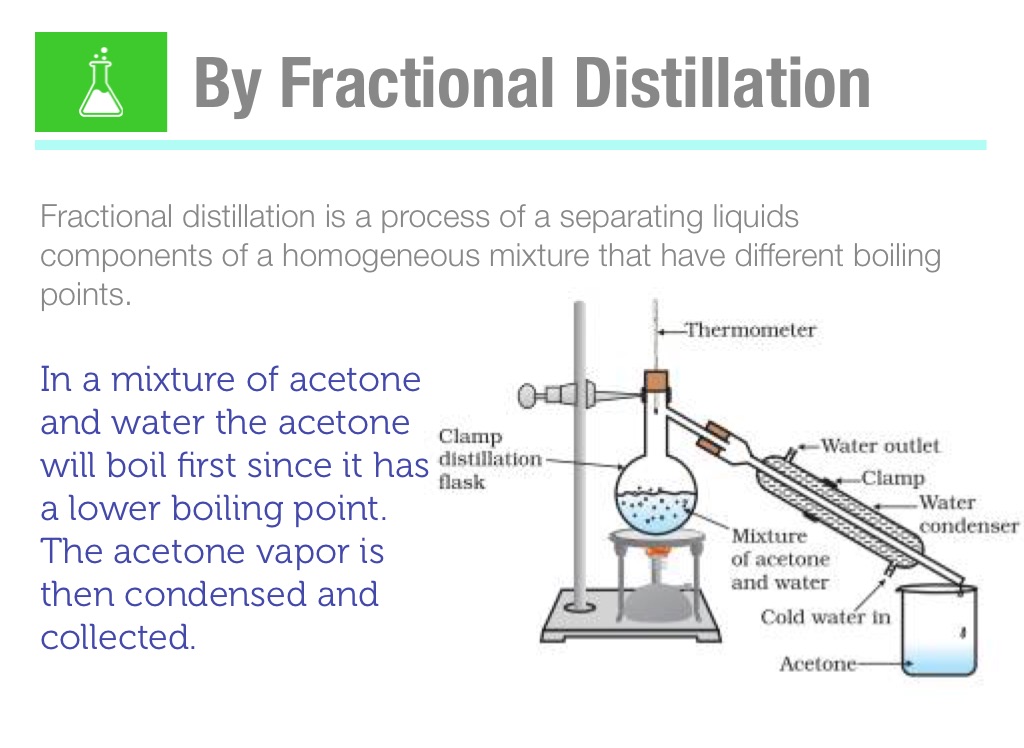
Simple Distillation 120 100 80 60 40 20 0 0 5 10 15 20 25 30 35 40 2. Using the graph calculate the boiling points of ac - Answer Happy

The boiling point of pure acetone is 56.38^(@)C. When 0.707 g of a compound is dissolved in 10 g of acetone there is elevation to 56.88^(@)C in b.p What is the mol.wt.

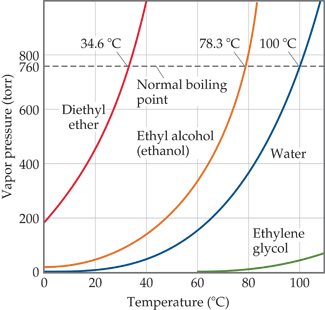
SOLVED:Question 5: (20 marks) The normal boiling point of acetone, an important laboratory and industrial solvent; is 56.2 "C and its AHvap is 25.5 kJ mol-!. At what temperature does acetone have

If the normal boiling point of acetone is 56^∘ C and it has a Δ H^∘vap of 32.1 kJ/mol, estimate the boiling point at 5 bar?

calculate the boiling point of a solution containing 045g of camphor mol wt 152 dissolved in 354g of acetone bp 563 c kb per 100 gm of acetone is 172 - Chemistry - TopperLearning.com | 9404yr66